We’re sharing everything you need to know about USDA APHIS VS Guidance 15201.1 and how it affects practitioners and labs performing EIA testing, as well as helpful resources to learn more about the changing requirements.
New EIA requirements take effect April 15. GVL is ready.
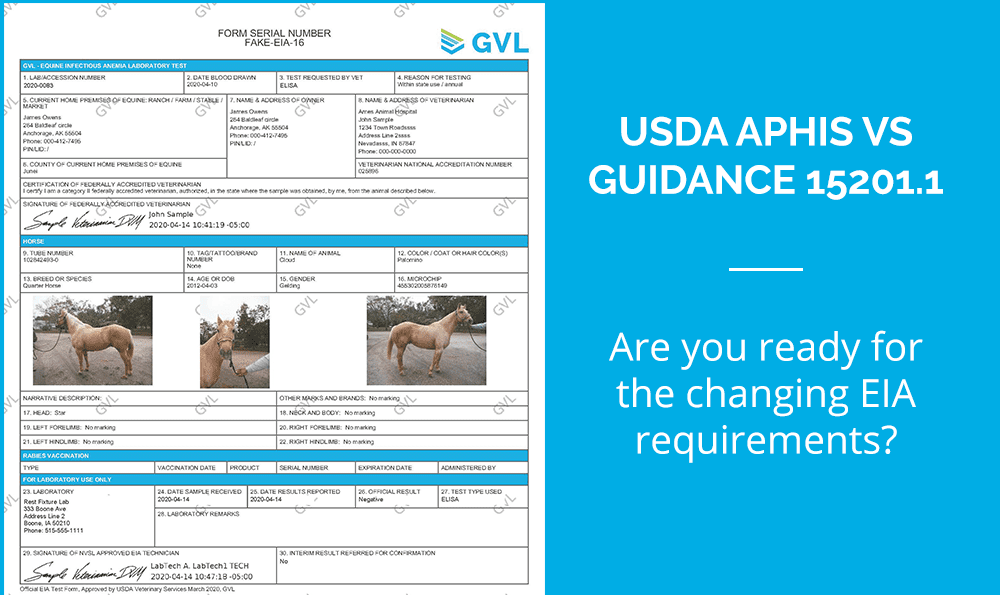
The USDA APHIS Veterinary Service Guidance 15201.1 – Approval of Laboratories to Conduct Tests for Equine Infectious Anemia requires all EIA test forms to be approved by the USDA for use by practitioners and laboratories by April 15, 2020 to be compliant.
After working directly with the USDA to ensure compliance, we have made several updates to the GVL platform and our EIA test certificates.
We received USDA approval and now includes the following statement on all EIA tests: “Official EIA Test Form, Approved by USDA Veterinary Services March 2020, GVL.”
“Our goal is to make it simple for veterinarians to comply with changing regulations,” said GVL Product Manager Michaela Whitney. “We’ve been aware of the new EIA guidance for some time and have worked diligently to ensure that every aspect of our test forms meet the new requirements.”
Learn more in our EIA help center
Explore our EIA help center, where we address common questions and include links to the USDA APHIS VS guidance resources.
- What changed about GVL EIA tests? We’ve made several updates to our EIA test forms to meet the new requirements for USDA approval.
- How can a veterinarian check their accreditation status? With this EIA guidance update, laboratories can only accept samples from federally authorized Cat II Accredited veterinarians.
- What does the guidance update for EIA tests? There are several other changes to EIA requirements that affect veterinarians and laboratories.
- EIA Test Updates – References. Learn more about VS Guidance 15201.1 and see resources from the USDA.
National Veterinary Services Laboratories (NVSL)
The USDA also now requires reporting of non negative EIA test results directly to NVSL. We are working closely with NVSL to incorporate EIA confirmatory testing into the GVL platform. Our goal is to have non negative EIA tests automatically sync to the NVSL system for analysis.
EIA tests in the GVL platform
The GVL platform streamlines EIA test processing for veterinarians, laboratories, and state/federal regulators, resulting in faster turn-around time for tests referred for confirmation.
GVL provides a software platform for veterinarians, laboratories, state and federal regulators for digital EIA (“Coggins”) test submission and resulting, as well as other animal health records.